
When someone is diagnosed with multiple myeloma, the focus often turns to chemotherapy, stem cell transplants, or targeted drugs. But one of the most painful and life-altering parts of the disease doesn’t even show up in blood tests - it’s the bone disease. Over 80% of patients develop severe bone damage, and for many, it’s the reason they can’t walk, sleep, or even sit up without agony. This isn’t just about weak bones. It’s about bone destruction that outpaces healing, turning the skeleton into a battlefield where cancer cells and bone cells fight a losing war - and the patient pays the price.
How Myeloma Turns Bone Into Swiss Cheese
Your bones aren’t static. They’re alive, constantly being broken down and rebuilt. Osteoclasts chew up old bone. Osteoblasts lay down new bone. In a healthy person, these two processes balance out. In multiple myeloma, that balance shatters.Myeloma cells don’t just sit in the bone marrow - they hijack the bone’s natural repair system. They pump out chemicals like RANKL, which tells osteoclasts to go wild. At the same time, they release DKK1 and sclerostin - proteins that shut down osteoblasts completely. No new bone forms. Meanwhile, osteoclasts chew away at existing bone, creating those classic "punched-out" holes you see on X-rays.
This isn’t random. The damage is localized. Bone biopsies show osteoclasts clustered right next to myeloma cells. Every time a piece of bone is destroyed, it releases growth factors trapped in the bone matrix - factors that feed the myeloma cells. So, bone destruction doesn’t just happen because of cancer. It fuels the cancer. It’s a vicious cycle: more tumor → more bone loss → more tumor.
Even osteocytes - the most abundant bone cells, making up 95% of bone tissue - get pulled into the fight. Studies show these cells start signaling differently in myeloma, releasing more sclerostin and worsening the imbalance. It’s not just the tumor. It’s the whole bone environment turning against itself.
The Cost of Broken Bones
Bone disease in myeloma isn’t just about pain. It’s about life-altering complications.- Pathological fractures occur in 28-38% of patients - bones breaking from normal stress, like lifting a grocery bag or even coughing.
- Spinal cord compression affects 5-10%, potentially leading to paralysis if not treated fast.
- Hypercalcemia (high calcium from leaking bone) hits 25-30%, causing confusion, nausea, and kidney failure.
These aren’t rare side effects. They’re the norm. Bone complications are the second most common reason myeloma patients end up in the hospital - right after infections. The average hospital stay? Over eight days. And for many, the pain doesn’t go away even after treatment.
One patient on the Myeloma Crowd Reddit forum shared: "I’ve been on zoledronic acid for two years. My pain is still at 7/10. I can’t sleep on my side. I don’t trust my spine anymore." That’s not an outlier. In a 2022 survey of over 1,200 patients, 68% still had persistent bone pain despite standard therapy.
Current Treatments: Stopping the Leak, Not Fixing the Hole
For decades, the only tools we had were bisphosphonates - drugs like zoledronic acid and pamidronate. They slow down osteoclasts. They reduce fractures by about 15-18%. But they don’t rebuild bone. They just slow the destruction.Then came denosumab. This drug blocks RANKL, the main signal that tells osteoclasts to destroy bone. In trials, it was slightly better than bisphosphonates at preventing fractures. It’s given as a simple monthly shot under the skin - no IV, no hospital visit. That’s why 74% of patients in a 2021 Mayo Clinic study preferred it.
But here’s the catch: neither bisphosphonates nor denosumab heal bone. They just stop it from getting worse. And they come with risks. Bisphosphonates can hurt your kidneys - 22% of patients need dose changes because their creatinine clearance drops. Denosumab can cause severe low calcium - 18.5% of patients need supplements and monitoring. And then there’s MRONJ - medication-related osteonecrosis of the jaw. About 42% of patients on long-term therapy develop this painful condition where the jawbone starts dying. Dental work becomes risky. Many patients end up losing teeth.
The New Hope: Drugs That Don’t Just Stop Destruction - They Build Bone
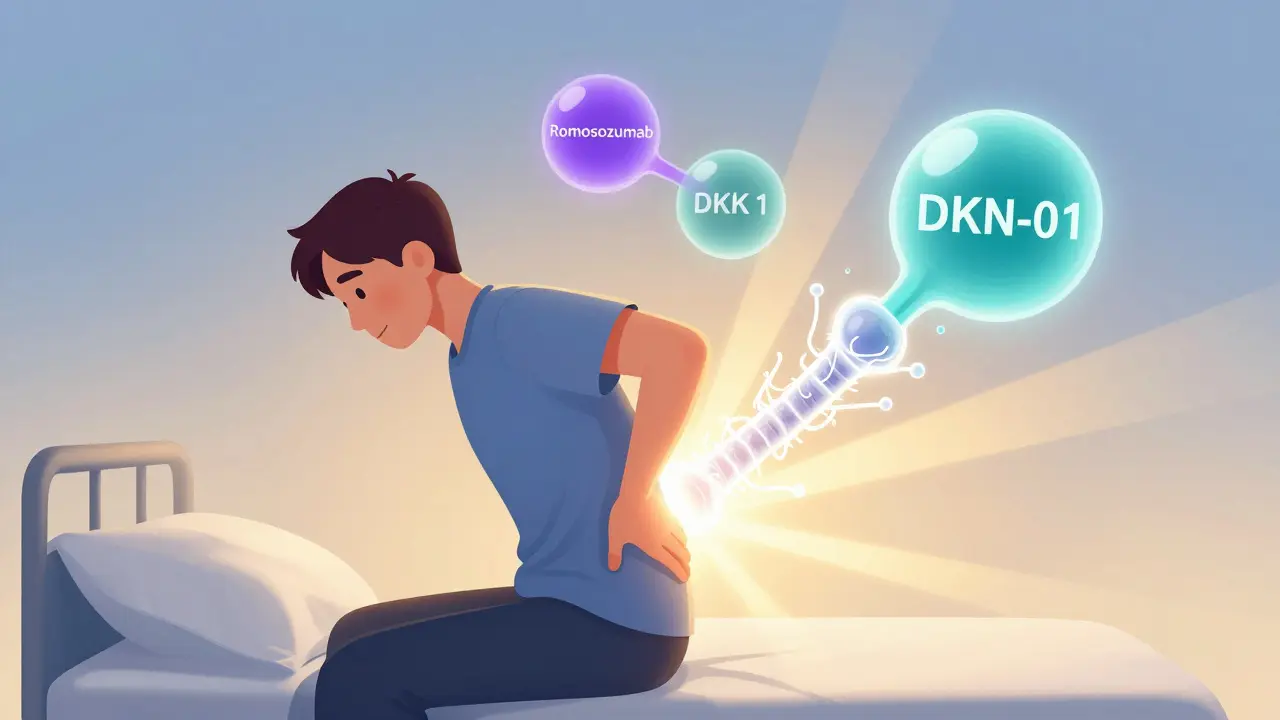
The real game-changer isn’t just stopping bone loss. It’s making bone grow again.One of the most exciting new drugs is romosozumab. It blocks sclerostin - the protein myeloma cells use to shut down osteoblasts. In a 2021 trial with 49 patients, romosozumab increased bone density in the spine by 53% in just 12 months. Patients reported a 35% drop in pain scores. This isn’t just slowing damage. It’s healing.
Another promising agent is DKN-01, which targets DKK1. In a 2020 trial with 32 patients, it cut bone resorption markers by 38%. That’s a direct hit on the main signal myeloma cells use to kill bone.
Even more exciting? Drugs that go after the Notch pathway. Myeloma cells use Notch3 and Notch4 to talk to bone cells and crank up bone destruction. Early trials with drugs like nirogacestat showed a 62% reduction in bone lesions in animal models. Human trials are just starting.
But not all new drugs made it. Odanacatib, a cathepsin K inhibitor, looked great in trials - 31% drop in bone breakdown. But it was pulled in 2016 because it raised stroke risk. That’s the lesson here: bone healing is powerful, but safety can’t be ignored.
Why We Still Can’t Cure Bone Disease
Despite all this progress, there’s a painful truth: no novel agent has yet proven it can extend survival. That’s not for lack of trying. Dr. Kenneth Anderson from Dana-Farber put it bluntly: "We’ve seen bone markers improve dramatically, but we haven’t seen patients live longer. We’re missing something. Maybe we need better ways to pick who will benefit."And that’s the next frontier. Not just giving drugs to everyone. But finding out who will respond. Who has high DKK1? Who has sclerostin levels over 28.7 pmol/L? Who’s at risk for fractures before they happen? The goal isn’t just to treat bone disease. It’s to predict it.
Right now, the International Myeloma Working Group says every newly diagnosed patient should get a full-body low-dose CT scan. That’s smart. But what if we could scan bone turnover markers monthly? What if we could tailor therapy based on your unique bone biology? That’s where the future is headed.
What’s Next? Healing, Not Just Managing
By 2028, the global market for myeloma bone disease drugs is expected to hit $5.1 billion. But money isn’t the point. The point is this: we’re moving from a world where we only prevent bone damage to one where we actually repair it.The FDA just approved a new, kidney-friendly version of zoledronic acid. The "BONE-HEAL" trial is now enrolling 450 patients to test romosozumab. RNA therapies are being tested to silence DKK1 at the genetic level. And bispecific antibodies - drugs that can target both myeloma cells and bone signals at once - are in early trials.
Dr. Brian Durie from the International Myeloma Foundation predicts that by 2030, we’ll stop seeing skeletal events as a major problem. Not because we’ve made myeloma disappear. But because we’ve made bone heal.
That’s the future. Not just surviving. But walking again. Sitting without pain. Lifting your grandchild without fear.
What causes bone damage in multiple myeloma?
Bone damage in multiple myeloma is caused by an imbalance between bone breakdown and bone formation. Myeloma cells release chemicals like RANKL and DKK1 that overactivate bone-destroying cells (osteoclasts) and shut down bone-building cells (osteoblasts). This leads to "punched-out" holes in the bone, called osteolytic lesions. The destruction also releases growth factors that feed the cancer, creating a vicious cycle.
What are the most common bone complications in multiple myeloma?
The most common complications are pathological fractures (28-38% of patients), spinal cord compression (5-10%), and hypercalcemia (25-30%). These can lead to severe pain, paralysis, kidney failure, and hospitalization. Bone disease is the second most common reason for hospitalization in myeloma patients, after infections.
What drugs are currently used to treat myeloma bone disease?
The standard treatments are bisphosphonates (zoledronic acid or pamidronate) and denosumab. Bisphosphonates are given as monthly IV infusions and can harm kidney function. Denosumab is a monthly injection under the skin that blocks RANKL, a key driver of bone destruction. Both reduce fractures but do not rebuild bone.
Do current bone drugs heal damaged bone?
No. Current drugs like bisphosphonates and denosumab only slow bone destruction. They do not stimulate new bone growth. This is a major unmet need. New drugs like romosozumab and anti-DKK1 therapies are being tested to actually rebuild bone, not just prevent further loss.
What are the risks of bone disease treatments?
Bisphosphonates can cause kidney damage, especially in patients with reduced kidney function. Denosumab can lead to low calcium levels (hypocalcemia), requiring calcium and vitamin D supplements. Both can cause medication-related osteonecrosis of the jaw (MRONJ), a condition where jawbone tissue dies, often requiring dental surgery. Patients should have a dental exam before starting treatment.
Are there any new drugs in development for myeloma bone disease?
Yes. Romosozumab (anti-sclerostin) increased bone density by 53% in a 2021 trial. DKN-01 (anti-DKK1) reduced bone resorption by 38%. Nirogacestat (anti-Notch) cut bone lesions by 62% in animal studies. RNA therapies like ALN-DKK1 are also being tested to silence bone-damaging signals at the genetic level. These are still in clinical trials but represent the next generation of treatment.




Kal Lambert
March 17, 2026 AT 12:43Just want to say: the fact that we're finally moving from 'slow the damage' to 'heal the bone' is huge. For years, we treated bone disease like a side effect, not the core problem. Romosozumab data? That’s not just promising-it’s paradigm-shifting. Patients aren’t just living longer. They’re living better. Walking. Sitting. Lifting grandkids. That’s the win.
And yes, we still need better biomarkers to predict who’ll respond. But we’re getting there.
Emily Hager
March 19, 2026 AT 11:42While I appreciate the optimism, I must emphasize that the data presented here lacks the rigor required for clinical adoption. The sample sizes are minuscule-49 patients for romosozumab? 32 for DKN-01? These are not statistically significant trials. Furthermore, the absence of long-term survival data renders these findings clinically irrelevant. One cannot base therapeutic paradigms on pilot studies with no control groups. This is premature enthusiasm masquerading as science.
Melissa Starks
March 20, 2026 AT 01:41I’ve been on denosumab for 3 years and I still can’t sleep on my side. My spine feels like it’s made of glass. I don’t care how fancy the new drugs are-if I’m still in pain after all this, then what’s the point? They talk about healing bone like it’s some magic fix, but no one talks about the jaw necrosis, the calcium crashes, the fact that you can’t get a tooth pulled without risking your life.
And don’t get me started on the cost. My insurance denied romosozumab because it’s ‘not first-line.’ First-line? I’ve been first-line for 7 years and I’m still broken. We’re treating the disease, not the person. And the person? The person is just… tired.
Lauren Volpi
March 20, 2026 AT 04:38So we’re spending billions on drugs that ‘heal bone’ but can’t cure cancer? LOL. This is why medicine is broken. You fix the symptom, not the cause. Meanwhile, real solutions-like lifestyle, fasting, turmeric, oxygen therapy-are ignored because Big Pharma doesn’t profit from them. Bone disease? It’s just a distraction. The real problem is the immune system collapsing. But hey, let’s keep injecting people with expensive biologics while they starve on $10K/month bills.
Ayan Khan
March 21, 2026 AT 22:46There is a quiet dignity in the way bone tissue fights back-even as it is torn apart. The osteocytes, those ancient sentinels of the skeleton, do not surrender. They signal, they cry out through sclerostin, not out of malice, but because they are trapped in a system they did not choose. This is not merely a disease of cells. It is a tragedy of communication. The tumor speaks a language the bone cannot refuse. And we, as healers, have spent decades trying to silence the destroyer, while neglecting the voice of the builder.
Perhaps the real breakthrough lies not in more drugs, but in listening-to the bone, to the patient, to the silence between the scans. Healing is not just chemical. It is relational. It is ethical. It is human.
Melissa Stansbury
March 23, 2026 AT 18:08Just read this whole thing and I’m crying. Not because it’s sad, but because it’s true. I had a compression fracture after sneezing. I was 52. I thought I was in shape. Turns out, my spine was Swiss cheese. I’ve been on zoledronic acid since diagnosis. My kidneys are fine, but my jaw? I lost two teeth. I didn’t even know that was a thing until it happened.
And now they’re saying we can rebuild bone? Like… actually rebuild it? I don’t know what to feel. Hope? Fear? Both? I just want to sit on the floor with my dog again without thinking, ‘Will this break?’
cara s
March 23, 2026 AT 23:51While the scientific narrative presented here is compelling, one must consider the epistemological framework underpinning these claims. The reductionist model of bone disease-as a mere imbalance between osteoclasts and osteoblasts-fails to account for the systemic, bio-energetic dysregulation inherent in neoplastic microenvironments. The emphasis on pharmacological intervention, while pragmatic, may obscure deeper metabolic and immunological perturbations. Furthermore, the reliance on biomarkers such as DKK1 and sclerostin as diagnostic proxies risks reifying proxy variables as causal agents. A truly integrative approach would require systems biology, not single-pathway inhibition. The future of myeloma bone disease lies not in more drugs, but in more holistic understanding.
Amadi Kenneth
March 24, 2026 AT 19:46Wait-so you’re telling me the government and Big Pharma are hiding the real cure? I’ve been researching this for months. Bone disease? It’s all about fluoride in the water. And 5G. They use the myeloma patients to test the EMF weapons. That’s why the bones turn to dust. They don’t want you to know that calcium supplements don’t work because the radiation blocks absorption. The CT scans? They’re tracking your bone density so they can target you with the next phase. I’ve got 30 years of research. I’ve sent letters to the WHO. No one answers. But I know. I know.